Bioquell Qlear
Integrated & automated hydrogen peroxide vapour bio-decontamination for critical areas in your facility. Deliver a bio-decontamination cycle with a 6-log sporicidal kill on exposed surfaces in a single zone or up to 8 selected distribution points. Learn more.
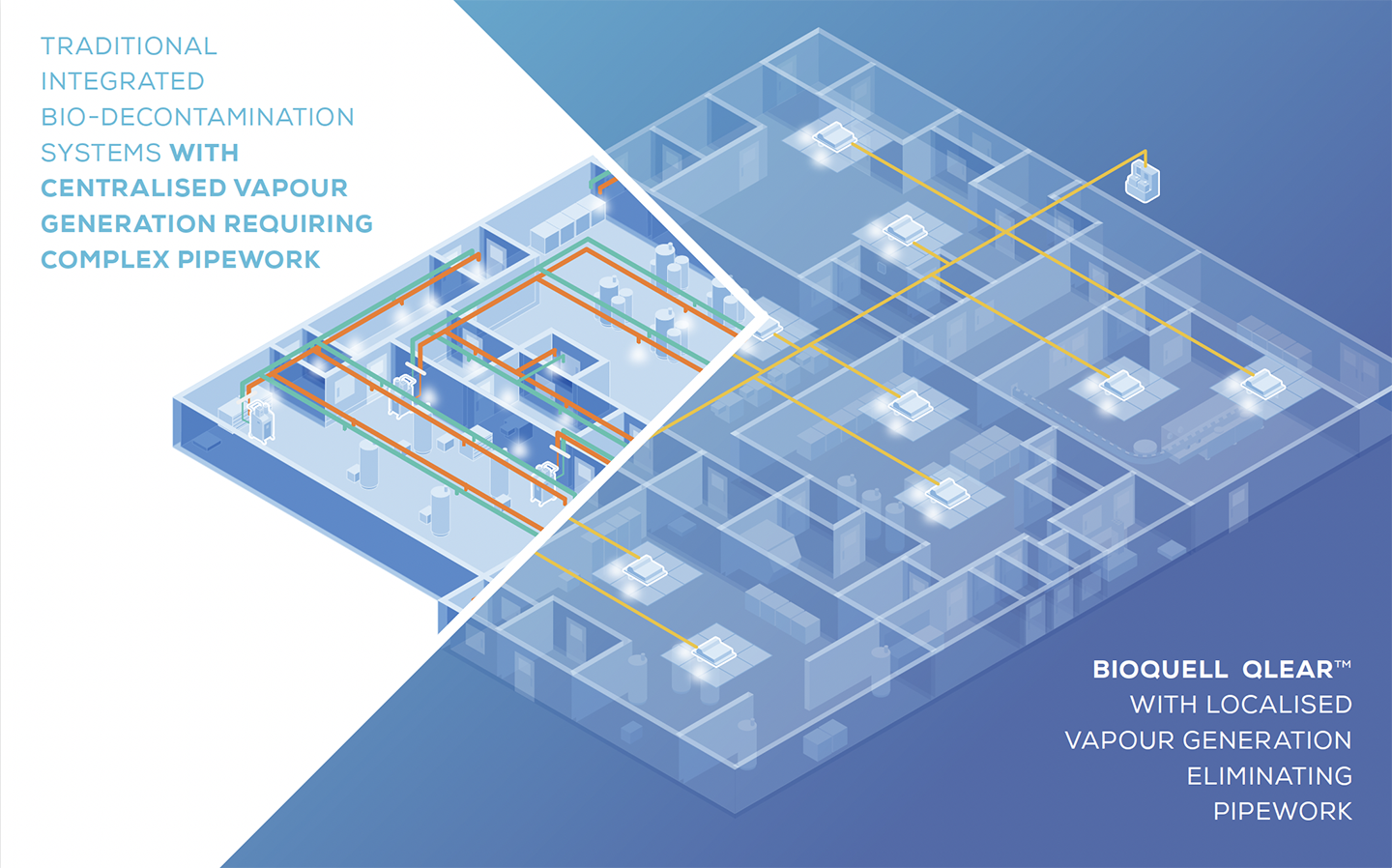
ELIMINATE THE NEED FOR COMPLEX AND COSTLY
PIPEWORK OR INCONSISTENT HVAC DISTRIBUTION
BENEFITS OF BIOQUELL QLEAR™




COMPONENTS OF BIOQUELL QLEAR™
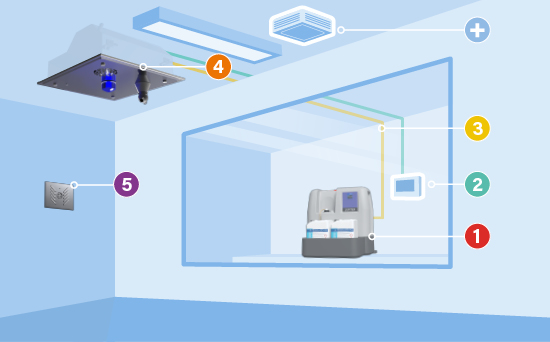
Components of Bioquell Qlear™
1. The hydrogen peroxide liquid station
2. The control panel
3. Flexible tubing
4. Vaporiser units
5. Environmental monitoring unit
+. Optional aeration units
HOW BIOQUELL QLEAR™ WORKS

FASTER INSTALL & FASTER CYCLE TIMES*
WITH NO EQUIPMENT SETUP NEEDED

Download a copy of the brochure to learn about our new modular integrated system that provides automated hydrogen peroxide vapour bio-decontamination for critical areas in your facility.
*Compared to alternative integrated bio-decontamination systems
BIOQUELL QLEAR™ DELIVERS
Streamlined Implementation
Local vapour generation reduces risk of validation failures and significantly shortens installation time
Ease of Use
Bioquell Qlear provides fully automated biodecontamination cycles at the touch of a button, eliminating equipment setup, disassembly and storage needs
Seamless Adaptability
Capable of bio-decontaminating individual rooms or complete zones while integrating seamlessly with building management systems (BMS) to ensure safe operation
COMPREHENSIVE SOLUTIONS FOR CLEANING,
DISINFECTION AND BIO-DECONTAMINATION

Effective manual cleaning and disinfection
Ecolab’s Klercide range of cleaning and disinfection products can be confidently used on the surfaces of the Bioquell Qube and other surfaces in the cleanroom.
Automated bio-decontamination systems for rooms, enclosures and equipment
A full range of Hydrogen Peroxide Vapour bio-decontamination solutions that help eliminate microorganisms throughout your process, from rooms to pass-throughs to existing isolators and more.

Correction action and emergency response capabilities
With Bioquell Rapid Bio Decontamination Service (RBDS), an expert rapid response team can be deployed to meet scheduled, or emergency needs of facilities across the globe providing professional and validated bio-decontamination services.
FAQs
Will this offer 21-CFR Part 11 compliance?
Yes, we are able to offer an audit trail software package to support compliance with 21 CFR Part 11.
What is the internal floor of the Bioquell Qube made from?
The floor of the Bioquell Qube is constructed from 316 stainless steel to ensure resistance to scratching on the working surface.
How can I bring tubing or cables into the Bioquell Qube?
We have an option for fitting a floor-mounted one-inch triclover port in each chamber. This allows you to bring in liquid lines or cables securely into the body of the Bioquell Qube.
What sets the Bioquell Qube apart from traditional stainless steel options?
The Bioquell Qube is constructed of tough and hard-wearing polypropylene, which is resistant to most materials the unit is ever likely to come into contact with. Because of its construction, the Bioquell Qube can be installed and validated within 16 weeks and, in most cases with minimal invasiveness to your operation or workflow.
Can you set alarms for the temperature inside the Bioquell Qube?
Yes.
If I wish to change pressure from positive to negative, do I need to change the filters or take any additional action to ensure it is safe to use?
The Bioquell Qube can be switched between pressure regimes simply with the touch screen; no additional changes are needed.
Are there different leak tests programmed for the Bioquell Qube?
Yes, there are leak tests programmed for both positive and negative pressure.
I have a small sterility-test need right now. What happens when my needs change?
The Bioquell Qube allows you to add and expand the system as your testing needs change and grow.
Contact Us
To learn more about how Bioquell can fit your solution, please contact us.
The Americas
Ecolab Inc
702 Electronic Dr. Suite 200
Horsham, PA 19044
+1 215 682 0225
bioquellusorders@ecolab.com





