Bioquell Qlear™
Integrated & automated hydrogen peroxide vapour bio-decontamination for critical areas in your facility. Deliver a bio-decontamination cycle with a 6-log sporicidal kill on exposed surfaces in a single zone or up to 8 selected distribution points. Learn more.
Warning: Undefined array key 0 in /bitnami/wordpress/wp-content/themes/Bioquell/temp-bioquell_qlear.php on line 69
ELIMINATE THE NEED FOR COMPLEX AND COSTLY
PIPEWORK OR INCONSISTENT HVAC DISTRIBUTION
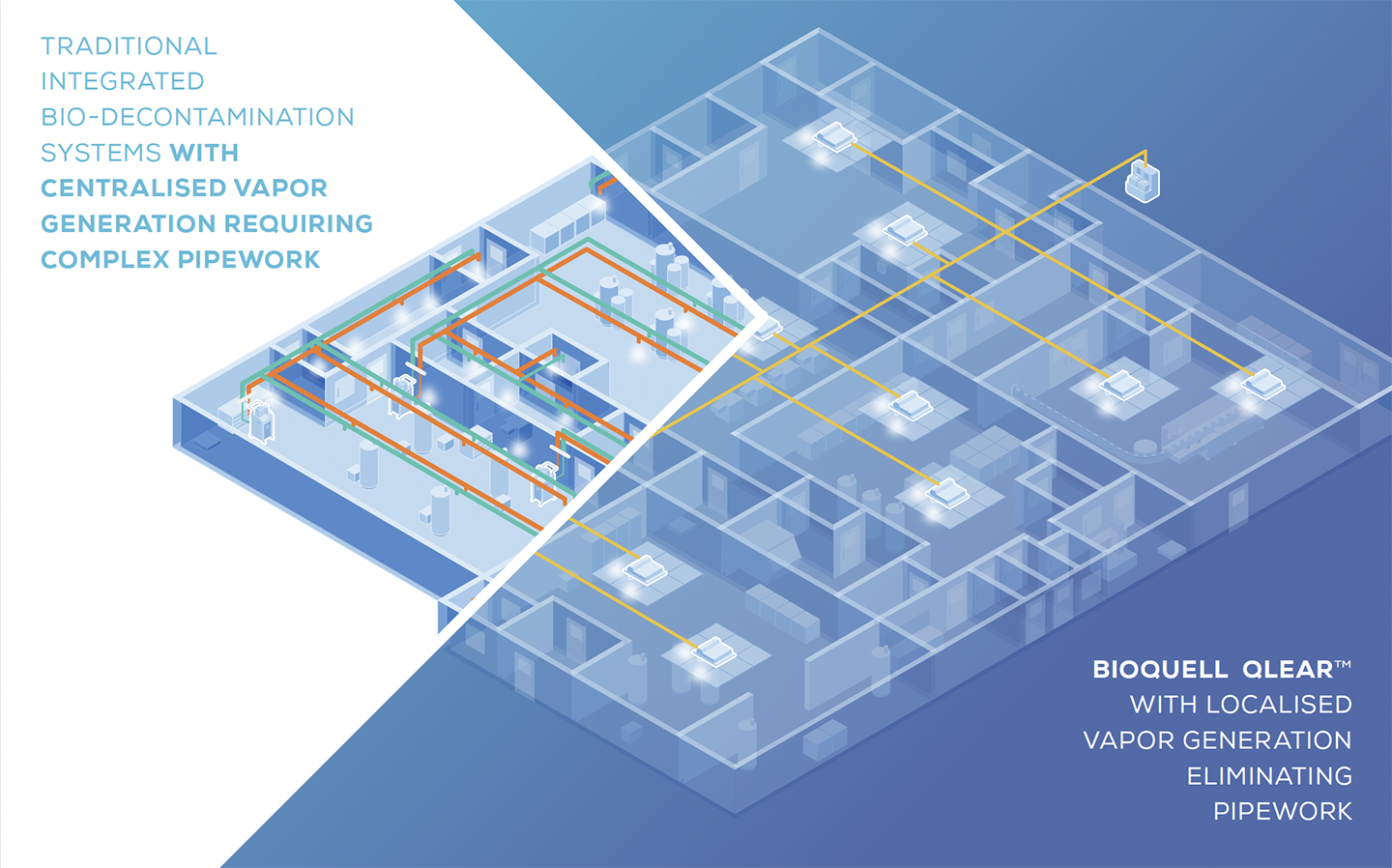
BENEFITS OF BIOQUELL QLEAR™




COMPONENTS OF BIOQUELL QLEAR™
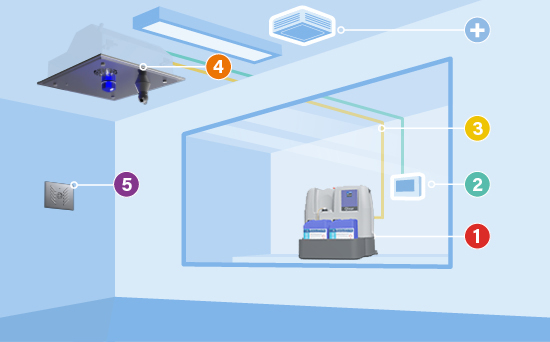
Components of Bioquell Qlear™
1. The hydrogen peroxide liquid station
2. The control panel
3. Flexible tubing
4. Vaporizer units
5. Environmental monitoring unit
+. Optional aeration units
HOW THE BIOQUELL QLEAR™ SYSTEM WORKS
Bioquell Qlear™ system in action
Watch the Video to Learn How the Bioquell Qlear™ System Works

FAST INSTALL & FASTER CYCLE TIMES*
WITH NO EQUIPMENT SETUP NEEDED

Download a copy of the brochure to learn about our new modular integrated system that provides automated hydrogen peroxide vapour bio-decontamination for critical areas in your facility.
*Compared to existing Bioquell bio-decontamination systems
BIOQUELL QLEAR™ SYSTEM DELIVERS
Streamlined Implementation
Local vapour generation reduces risk of validation failures and significantly shortens installation time
Ease of Use
Bioquell Qlear™ system provides fully automated bio-decontamination cycles at the touch of a button, eliminating equipment setup, disassembly and storage needs
Seamless Adaptability
Capable of bio-decontaminating individual rooms or complete zones while integrating seamlessly with building management systems (BMS) to ensure safe operation
COMPREHENSIVE SOLUTIONS FOR CLEANING,
DISINFECTION AND BIO-DECONTAMINATION

Effective manual cleaning and disinfection
Ecolab’s Klercide range of cleaning and disinfection products can be confidently used on the surfaces of the Bioquell Qube™ isolator and other surfaces in the cleanroom.
Automated bio-decontamination systems for rooms, enclosures and equipment
A full range of Hydrogen Peroxide Vapour bio-decontamination solutions that help eliminate microorganisms throughout your process, from rooms to pass-throughs to existing isolators and more.

Correction action and emergency response capabilities
With Bioquell Rapid Bio Decontamination Service (RBDS), an expert rapid response team can be deployed to meet scheduled, or emergency needs of facilities across the globe providing professional and validated bio-decontamination services.
FAQs
How does Bioquell Qlear™ integrated bio-decontamination work?
Unlike other integrated bio-decontamination systems which generate hydrogen peroxide vapor remotely and distribute it into rooms via HVAC ducting or rigid pipework, the Bioquell Qlear system distributes the hydrogen peroxide from a remote station through liquid tubes and the vapor is generated locally from vaporizer units that are permanently located within each target room. This simplifies installation, increases the repeatability of the bio-decontamination process, and helps reduce cycle times.
How many rooms can Bioquell Qlear™ system bio-decontaminate?
Nominally, each system can bio-decontaminate 1,000m3 of space. The number of rooms will depend on the size of the rooms, but if required, multiple vaporizer units can be installed into larger rooms to achieve a uniform and repeatable bio-decontamination process.
Can the Bioquell Qlear™ system bio-decontaminate individual rooms or do all rooms have to be treated together?
The Bioquell Qlear™ system is completely configurable with each system allowing for up to 8 separate programs or zones, meaning it is possible to bio-decontaminate rooms and material airlocks individually as required, or as an entire suite.
How does the Bioquell Qlear™ system ensure that the operator has selected the intended room for bio-decontamination?
Each zone is equipped with an in-room warning panel with a key switch that the operator must physically turn when going into the room to check that it is empty of personnel. The bio-decontamination cycle will not start unless the key switch for the specific zone selected has been activated.
How does the Bioquell Qlear™ system prevent leakage of hydrogen peroxide?
The tubing is made from a material that is resistant to peroxide. Ecolab also advise to have a protective conduit around the tubing to avoid any mechanical damage. The Bioquell Qlear™ system can also detect the presence of liquid at each vaporizer.
What inputs does the Bioquell Qlear™ system require?
The liquid station, control panel and vaporizers simply require a standard 230/120V power supply. Additionally, each vaporizer requires a standard RJ-45 cable to connect to the control panel, as well as a liquid tube connected to the liquid station.
How is the system installed?
Ecolab’s project management team will work seamlessly with the contractors responsible for building the facility where the Bioquell Qlear system is being installed to ensure that each party understands what they are responsible for. In general, the contractor will install all cabling and tubing and install the outer casings of each system component. Ecolab will then place make the final connections before commencing with commissioning and validation activities.
Is an integrated bio-decontamination system suitable for GMP pharmaceutical manufacturing?
Yes. The Bioquell Qlear integrated bio-decontamination system is designed to support GMP requirements by providing controlled, repeatable, and validated 6-log sporicidal kill on exposed surfaces. Bio-Decontamination systems are commonly installed in aseptic processing areas (including RABS), material airlocks, and cleanroom suites to support contamination control strategies.
Can integrated bio-decontamination support aseptic filling and sterile manufacturing areas?
The Bioquell Qlear integrated system is well suited for aseptic and sterile manufacturing environments because it delivers consistent, room-specific bio-decontamination without reliance on portable equipment which can be prone to human error if set up incorrectly. Integrating bio-decontamination equipment within the fabric of the cleanroom helps maintain cleanroom integrity and supports contamination control in critical environments.
How does an integrated system support contamination control strategies (CCS)?
By providing automated, documented, and repeatable bio-decontamination cycles, the Bioquell Qlear integrated system forms a key part of a facility’s contamination control strategy, particularly to address the need for a routine sporicidal disinfection. It supports routine, event-driven, and campaign-based bio-decontamination while reducing variability associated with manual disinfection processes.
How is an integrated bio-decontamination system validated in a pharmaceutical facility?
Validation typically includes installation qualification (IQ), operational qualification (OQ) to ensure that the system is operating as designed. This is followed by gassing cycle development to establish which parameters are required to achieve a repeatable 6-log sporicidal kill, followed by performance qualification (PQ) to confirm that the developed cycle is robust. The system is then validated for routine use. Integrated bio-decontamination systems support validation by eliminating manual equipment setup and consistent vapor distribution, simplifying ongoing requalification.
Warning: Undefined array key 11 in /bitnami/wordpress/wp-content/themes/Bioquell/temp-bioquell_qlear.php on line 463
Warning: Undefined array key 11 in /bitnami/wordpress/wp-content/themes/Bioquell/temp-bioquell_qlear.php on line 464
Contact Us
To learn more about how Bioquell can fit your solution, please contact us.
The Americas
Ecolab Inc
702 Electronic Dr. Suite 200
Horsham, PA 19044
+1 215 682 0225
bioquellusorders@ecolab.com
Global Offices
Deprecated: addslashes(): Passing null to parameter #1 ($string) of type string is deprecated in /bitnami/wordpress/wp-content/themes/Bioquell/functions.php on line 530





